An earlier post touched briefly on the use of nuclear imaging in clinical trials (with a later one zoning in on PET). Here, we focus on why and how imaging is used to select patients for clinical trials.
Why Select Patients?
It is well known that patients respond to therapeutics in diverse ways. Nowadays, drug development is increasingly heading towards the direction of ‘personalized’ or ‘precision’ medicine. According to one report, this means “using molecular profiling technologies for tailoring the right therapeutic strategy for the right person at the right time, and determine the predisposition to disease at the population level and to deliver timely and stratified prevention”.
Oncology has seen the most success regarding this approach, with insights into the molecular and genetic make-up of cancer translating into highly effective therapies. These drugs target specific factors that are linked to the development, progression, and spread of cancer, such as the estrogen receptor (tamoxifen, letrozole), the BCR-ABL fusion protein kinase (imatinib), and vascular epidermal growth factor (bevacizumab).
How to Apply Imaging to Select Patients
For imaging CROs, imaging for patient selection is based on eligibility criteria (see Box 1 for a definition). For cancer, such criteria might include minimum tumor number/size at baseline or the expression of specific molecules on the lesions in case of a targeted therapy. Indeed, with the growth of therapeutics that act on specific biological molecular targets, a parallel increase has occurred regarding the development of imaging biomarkers (see Box 1 for a definition).
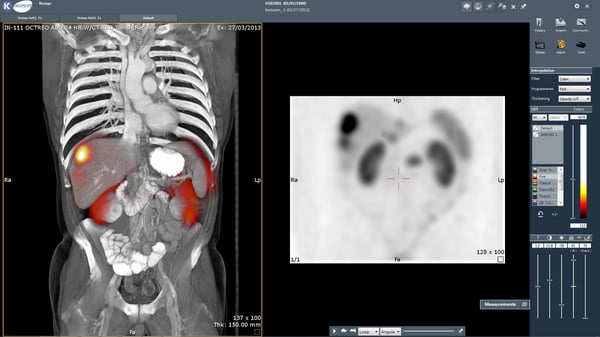
If an imaging biomarker is used to select patients, then close collaboration between the radiologists (who identify the tumors) and nuclear physicians (who verify that that tumor indeed expresses the required molecule) is fundamental.
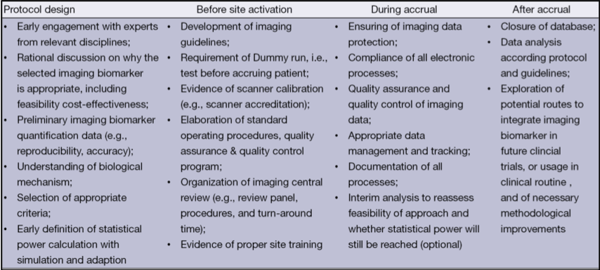
Many other aspects require careful consideration when applying imaging to select patients for trials (see Fig. 1). For example, even before the trial begins, it must be assured that the imaging biomarker is fit for purpose. Once that has been proven, then developing imaging guidelines and applying them consistently is crucial. Getting these steps right will ensure that the right patients are receiving the therapy.
Imaging for Patient Selection: Beyond Oncology
Advances in imaging capabilities and a growing number of radiotracers are expanding the range of diseases that can benefit from using imaging to select patients for trials. Here, a recent example for a neurological indication, a field that is notoriously difficult to develop disease-modifying drugs for, is discussed.
Last year, the European Medicines Agency (EMA) issued a positive qualification opinion on an imaging biomarker as a tool to enrich Parkinson’s clinical trials. The radiotracer 123I-ioflupane binds specifically to dopamine transporters at sites on the neurons that are lost in Parkinson’s disease and helps in diagnosis. As with many neurodegenerative diseases, giving treatment as early as possible is optimal to slow down or stop pathological changes. However, signs and symptoms of early Parkinson’s are usually mild, making it challenging to identify eligible patients to enrol in trials. This qualified biomarker (detected via SPECT) provides a vital tool in overcoming this barrier.
As stated by the lead scientist driving this project, this tracer is not new but there was a lack of clarity regarding whether it could be used to select patients for clinical trials. Through a global effort to compile a compelling case, “This endorsement from the European Medicines Agency represents many years of hard work and incredible collaboration among companies, universities, and charities facilitated by the Critical Path Institute.”
Final Thoughts
Very few imaging biomarkers are validated/qualified by regulatory agencies. Indeed, validation/qualification can take years of effort among diverse stakeholders, as exemplified by 123I-ioflupane. Moreover, if an imaging biomarker is essential for selecting patients for a clinical trial, it may then become a regulatory requirement for when the drug is approved. That is, it may become a companion diagnostic. Considerations are therefore needed regarding the type of data required for submission to the regulatory authorities.
There are many challenges to using imaging for patient selection (see Fig. 1 and Table 1); however, this approach is playing an increasingly vital role in helping reduce the time needed for drug approval.
Eligibility Criteria: standards listed in the protocol outlining who can participate within a given clinical trial. Patients are selected to participate in the study according to these criteria.
Imaging Biomarker: a biologic feature detectable in an image. However, this term is used in different contexts and may be very confusing. For example, an imaging biomarker can be a measurable variable that is a (surrogate) endpoint for the trial, such as RECIST 1.1 for tumors. For patient selection, a biomarker can be used to detect the presence or absence of a molecular target, e.g., a specific receptor, before enrollment into a trial.